INTRODUCTION
Hemoperitoneum is an important sign of deterioration in the thoraco-abdominal trauma which requires in the majority of cases urgent surgical intervention. Even if mild, any trauma of the abdominal wall can be followed by some type of internal organ damage. Early diagnosis and treatment may be extremely important in saving patient’s life1.
Acute gastric infarction is rare condition primarily due to the rich vascularization of the stomach comprising the right and left gastric arteries as well as the right and left gastroepiploic arteries. As a consequence, there are only few cases of gastric infarction reported in the literature. This event may be determined by vascular, chemical, mechanical or infectious etiologies2. The first case of a major gastric infarction was reported in 1909 by Baumann3. He described it as an accidental event at an autopsy of a 90-year-old woman. A surface of approximately 10 cm was hemorrhagic and spreading along a 2 cm surface above the pyloric sphincter. The left and right gastric arteries as well as the right gastroepiploic artery and also some of the biggest derived branches had been blocked by an adherent thrombus4.
The onset of symptoms is generally catastrophic and, therefore, surgical intervention must be urgently employed because of the steep increase in mortality due to delay of diagnosis and treatment5. The spleen is the most frequently affected solid organ in trauma suffering both from blunt or penetrating lesions. It is rather common that penetrating trauma result in significant damage6. Several classification systems based on the severity and localization of spleen trauma have been described and reviewed in Table 17,8.
Table 1
Spleen trauma classification
CASE PRESENTATION
We are presenting here the case of a patient aged 84 years who came to the ICU of the ‘Sf Ioan’ Clinical Emergency Hospital from Bucharest with unspecific clinical symptoms: confusion, agitation, tachycardia, hypotension and presenting Livedo reticularis. The personal medical history of the patient revealed that he was suffering from type 2 diabetes, arterial hypertension, coronary heart disease, stage III chronic kidney disease and deep vein thrombosis. It was also revealed that 2 weeks earlier he had suffered a same-height fall. During clinical examination abdominal distension, spontaneously pain and signs of peritoneal irritation on palpation and absence of bowel movement were revealed. After admission, the vital signs were continuously monitored and further paraclinical investigations were initiated. Following the protocol, a SARS-CoV-2 PCR test was also done. Following a CT scan without intravenous contrast medium of thorax, abdomen and pelvis that was performed in an emergency basis, an exploratory laparotomy of the peritoneal cavity was decided.
From the observation of the preoperative vital signs, arterial hypotension of 90/53 mmHg, with 110 b/min pulse and oxygen desaturation of 88% were recorded. The paraclinical examinations revealed: a severe anemia with a Hgb=7.2 g/dL and Hct=24%, thrombocytopenia (PLT=43000 U/μL), WBC=5800 U/μL, fibrinogen: 1200 mg/dL, urea=37 mg/dL and creatinine=3.04 mg/dL. The PCR SARS-CoV-2 test came positive. Inflammatory markers such as CRP (23 mg/dL) and ferritin (800 ng/mL) levels were increased compared to the reference intervals, which is consistent both with the ischemic and infectious acute status of the patient.
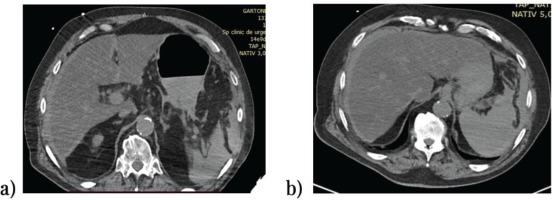
CT results revealed dense perihepatic, perigastric, interhepatogastric and perisplenic fluid collection, suggestive of hemoperitoneum (Figures 1a in longitudinal section as well as Figures 2a and 2b in transverse section) with gastric pneumatosis (Figure 1b in longitudinal sections), without any COVID-19 specific pulmonary lesions.
Figure 1
CT scan of thorax, abdomen and pelvis – longitudinal section: a) splenic hematoma with capsular rupture, and b) voluminous gastric aeric dilatation
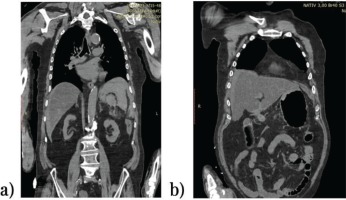
Figure 2
CT scan of abdomen, transverse section: a) splenic hematoma, and b) splenic and perisplenic hematoma
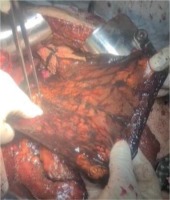
Considering all clinical and paraclinical results, it was decided to proceed with exploratory laparotomy. This intervention started with an exploratory laparotomy of the peritoneal cavity which revealed: hemoperitoneum with stage IV splenic trauma in 2 sites associated with gastric infarction with a quasi-total necrosis (Figure 3). It was later found that the celiac trunk branches, as well as the right gastric artery appeared to be unaffected intraoperatively.
As a consequence, damage control surgery was decided as the most appropriate solution. Based on this, splenectomy (Figure 4) associated with subtotal gastrectomy (Figure 5) were performed. Considering the hemodynamic instability of the patient as well as the precarious quality of the tissue which was improper for anastomosis, the gastric stump was closed and a feeding jejunostomy was performed.
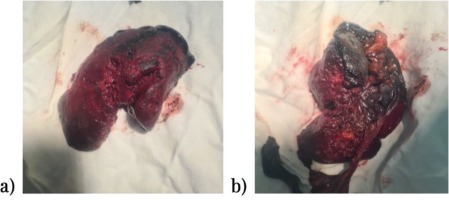
Figure 4
Postoperative image of spleen: a) splenic rupture, and b) the hematoma dissecting the splenic capsule completely
The postoperative course of the patient was unfavorable since post-operative cardiopulmonary arrest, unresponsive to resuscitation ultimately lead to his death. The histopathological examination revealed a gastric infarction with extensive transparietal necrosis and splenic hematoma, with no highlight of trombogenic evidence. Consequently, the gastric infarction occurred most probably due to splenic hematoma.
DISCUSSION
A small number of gastric infarctions has been described in literature9-14. Do et al.15 presented the case of a gastric ischemia after a gastric bypass surgery with a similar clinical presentation and end result to our case: A woman aged 63 years arrived to the ICU, presenting nausea, vomiting, diarrhea and worsening of diffuse abdominal pain. Her personal medical history included: arterial hypertension, type 2 diabetes, history of laparoscopic gastric bypass with Roux-en-Y reconstruction and cholecystectomy. No bowel perforation was identified during surgery, although focal gastric necrosis with purulent fluid collection in the peritoneal cavity was observed. The surgical procedure ended with the irrigation of the peritoneal cavity, the short gastric arteries ligation, subtotal small bowel resection and the remaking of Roux-en-Y gastrojejunostomy.
York et al.3 described a case of gastric necrosis with abdominal pain due to overfeeding, leading the patient to be admitted to the ICU. In this case, the authors describe that 2 L of serosanguineous exudate fluid from the stomach were extracted intraoperatively. The stomach was dilated, edematous, with a posterior wall gangrene. The partial gastrectomy and repair were done in 2 layers using Cornel and Lambert suture techniques3.
Perigela et al.2 described another case of gastric infarction of a patient with necrotizing gastritis. A man aged 60 years came to the ICU complaining of pain in the upper abdomen. The emergency exploratory laparotomy showed free fluid collection in the peritoneal cavity and gangrene in a proportion of 1/3 of the stomach with intact extrinsic blood supply. The microscopic examination of the stomach tissue revealed a full thickness necrosis of the wall in a diffuse necrotic process. Furthermore, after the exclusion of other causes it was suspected that the stomach was involved in a necrotizing infection. Taking into consideration the data collected from literature and the undeniable advantages of gastric Roux-en-Y bypass, this option becomes the obvious choice in case of gastric necrosis which requires subtotal or total gastrectomy, the remaking of the digestive continuity being ensured by a gastrojejunostomy or esophagojejunostomy accordingly2.
Other pathological examinations related to the cases of gastric infarction were communicated by Do et al.15 whose observations included transmural necrosis of the stomach wall with a certain submucosal hematoma. There were also found abscesses and mixed bacteria in the fibrinous exudate and necrotic tissue. Yorke et al.3 also showed that in their case the gastric wall presented transmural necrosis with congested blood vessels.
Related to the splenic pathology associated to the SARS-CoV-2 infection, in the literature there are described few cases of splenic rupture with hemoperitoneum on the background of SARS-CoV-2.
Mabaven et al.16 submitted the case of a patient aged 52 years who came to the emergency department (ED) with abdominal pains, fatigue, fever, nausea and a positive result of SARS-CoV-2 PRC test. The abdomen scan revealed an extended perisplenic fluid collection. An exploratory laparotomy was performed during which a large volume of blood was drained. After suction a splenectomy was performed. The cause of the hemoperitoneum was attributed to the atraumatic rupture of the spleen on the SARS-CoV-2 background.
Shaukat et al.17 depicted the case of a patient aged 57 years arriving at the ED in a deteriorated condition that started 10 days before. Dry cough, diarrhea and loss of appetite without trauma history were reported. Medical records included moderate obstructive sleep apnea and chronic bronchitis as well as the excision of inferior lobe of the left lung, as a result of trauma 20 years ago. An urgent CT scan of the thorax and abdomen was performed. The result outlined fluid accumulation in both subdiaphragmatic spaces, paracolic spaces and pelvis suggesting hemoperitoneum. An embolization of splenic artery was performed in order to stop the bleeding. The post-operative course of the patient was favorable, taking into consideration the SARS-CoV-2 infection also diagnosed through a PCR test.
Knefati et al.18 also presented the case of a patient aged 75 years that was SARS-CoV-2 positive who presented in the ED with acute abdominal pain on the left side. CT of the abdomen /pelvis revealed a subcapsular fluid collection measuring 32 mm in thickness, surrounding the spleen and indicating a subcapsular hematoma. The exploratory laparotomy showed the splenic rupture with hemoperitoneum and, consequently, an urgent splenectomy was done.
Considering the few similar cases available in the literature on this pathology mainly in times of COVID-19, we could attribute the splenic hemorrhage pathology to the SARS-CoV-2 infection19-21. However, in our particular case there are a few other suspicions related to the cause of the gastric infarction. One of the causes could be the celiac trunk thrombosis, despite that the preoperative CT performed at the ED did not describe its existence. The right gastric artery was also apparently unaffected intraoperatively. Another possible cause could be the compression of the splenic hematoma on the celiac trunk, however, extremely improbable due to the multiple sources of alternative vascularization of the stomach. Therefore, it is debatable how this splenic hematoma appeared: following SARS-CoV-2 infection and/or due to trauma.
CONCLUSION
Gastric infarction is a rare condition due to a rich vascularization of the stomach. However, spleen trauma or other conditions could lead to this event, which is a pathology of major emergency, with high rates of mortality. Despite the low number of gastric infarctions reported in the literature, in the COVID-19 era, several SARS-CoV-2 cases were presented, indicating this infection as a possible etiology and/or co-morbidity for gastric infarction. In our particular case, based on the investigations and procedures done in emergency, we can conclude that the splenic rupture was most likely caused by traumatic injury, possibly aggravated by the infection with SARS-CoV-2. However, we can also conclude that the overall negative evolution of the patient may have been significantly influenced by COVID-19.