INTRODUCTION
Tobacco smoking remains one of the foremost preventable causes of morbidity and mortality worldwide, contributing to a broad spectrum of chronic health conditions that disproportionately affect older adults1. Recent global estimates indicate that smoking causes millions of deaths annually and accelerates the onset and progression of diseases traditionally associated with aging, such as cardiovascular disease (CVD), chronic obstructive pulmonary disease (COPD), neurodegenerative disorders and various metabolic syndromes2. Aging is inherently a non-modifiable biological process; smoking, however, is a fully modifiable exposure.
The harmful effects of smoking on age-related biological processes have become increasingly evident through advances in molecular epidemiology. Cigarette smoke induces oxidative stress, mitochondrial dysfunction, DNA damage and systemic inflammation mechanisms that mirror or accelerate natural aging pathways3. Smoking induces epigenetic alterations that closely resemble molecular signatures of biological aging, including hypermethylation at polycomb-regulated CpG sites and hypomethylation at enhancer regions, patterns associated with age-related epigenomic drift and accelerated biological aging4. In addition, tobacco exposure is linked to dose-dependent leukocyte telomere shortening, reflecting cumulative oxidative stress and increased cellular turnover that promote replicative senescence and genomic instability5. Importantly, some of these epigenetic and telomeric changes may persist even after smoking cessation, suggesting long-lasting biological effects that extend beyond direct toxic exposure6. Biomarker-based aging tools such as epigenetic clocks further demonstrate that smokers exhibit greater biological age acceleration than non-smokers, even after controlling for chronological age4. Klopack et al.5 showed that lifetime smoking exposure was significantly associated with faster epigenetic aging trajectories and a higher risk of chronic disease in older adults5. Similarly, evidence shows that smoking contributes to telomere shortening, endothelial dysfunction, inflammaging and immune senescence, thereby promoting vulnerability to late-life diseases4.
However, the effects of smoking are not homogeneous across organ systems. In cardiovascular health, smoking remains one of the strongest modifiable predictors of CVD morbidity and mortality in older adults6. Large-scale cohort studies demonstrate that smoking amplifies atherosclerotic processes, increases thrombosis risk, and accelerates vascular aging, leading to coronary heart disease, heart failure, and stroke7. Importantly, even light or intermittent smoking in late life is associated with notable increases in cardiovascular mortality8. Smoking also interacts synergistically with age-related endothelial decline, contributing to arterial stiffness and impaired vascular repair capacity, conditions tightly linked to age-related cardiovascular diseases9. This dual burden of chronological aging and smoke-induced vascular aging underscores the need to conceptualize smoking not merely as a risk factor, but as an accelerant of the aging process itself.
The respiratory system offers another compelling example of smoking’s role in accelerating age-related decline. COPD, a leading cause of disability and death among older adults, is widely recognized as a condition of accelerated lung aging driven in large part by cigarette smoke10. The chronic inhalation of toxic particles induces structural remodeling, alveolar destruction, airway inflammation, and irreversible functional impairments, changes analogous to those in older lungs but occurring at a faster pace11. Barnes6 emphasized that cigarette smoke enhances oxidative burden and degrades anti-aging mechanisms in pulmonary tissue, positioning COPD as both a smoking-related disease and an archetype of accelerated aging12. Moreover, the interplay of smoking with immune senescence leads to heightened susceptibility to respiratory infections, pneumonia, and acute exacerbations, further reinforcing the classification of smoking-driven pulmonary decline as a distinct aging trajectory.
Neurological health is similarly impacted. Substantial evidence links smoking to accelerated cognitive decline, neurovascular injury, and increased risk of dementia, including Alzheimer’s disease13. Neuroimaging studies reveal structural and functional brain alterations among chronic smokers, such as reduced cortical thickness, impaired white-matter integrity, and diminished executive functioning14. The mechanistic pathways connecting smoking to neurological aging include oxidative damage to neurons, microvascular ischemia, impaired neurogenesis, and increased accumulation of neurotoxic proteins15. A 2020 systematic review showed that late-life smokers had a significantly higher risk for dementia compared with non-smokers, even after adjusting for age and comorbidities16. These findings highlight that smoking operates as an age-accelerating agent in the brain, contributing to neurodegenerative disease trajectories.
Despite these extensive insights from epidemiology, clinical science, and aging biology, the research addressing the intersection of smoking and age-related diseases remains conceptually fragmented. Studies often examine smoking’s effects within a single disease category, cardiovascular, respiratory, neurological, or metabolic, without integrating the broader aging-centric framework. The need for such a unifying framework is underscored by the fact that smoking is one of the few major determinants of aging-related disease that can be modified. Age cannot be changed, but smoking cessation at any stage of life can significantly reduce the risk of cardiovascular, respiratory and neurological disease, even in older adults17. Positioning smoking within an aging-based paradigm reframes prevention: the aim becomes not only reducing disease risk but slowing biological aging pathways themselves. This shift is especially relevant for global aging societies, where preventing or delaying age-related diseases is a strategic priority18.
A bibliometric mapping approach is uniquely suited to analyze aging acceleration due to smoking. Bibliometric analysis enables the systematic quantification of global research patterns, major scholarly contributors, citation networks, thematic clusters and conceptual evolution across the domains of smoking, aging and disease. Through co-citation, keyword co-occurrence, co-authorship and thematic evolution analyses, a bibliometric approach can reveal how knowledge structures are formed, where evidence converges or diverges and which research gaps persist. Importantly, bibliometric mapping allows us to examine how the scientific community has implicitly addressed the components of aging acceleration due to smoking and age-related diseases. By conducting a comprehensive bibliometric mapping of the smoking–aging–disease nexus, this study seeks to bridge fragmented research domains and provide an empirical foundation for future interdisciplinary investigations.
METHODS
Study design
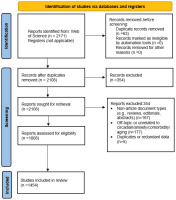
This study employed a quantitative bibliometric design to analyze global research patterns on smoking, aging, and age-related diseases. Although not a systematic review, the methodological procedures were structured in accordance with PRISMA 2020 guidelines (Figure 1).
Data source and search strategy
Data were retrieved exclusively from the Web of Science (WoS) Core Collection. The search was conducted in January 2025 using the following Topic (TS) query: TS = (‘smoking’ OR ‘tobacco’) AND (‘aging’ OR ‘age-related diseases’ OR ‘age related diseases’). The search covered publication years 2005–2025, yielding 2171 records.
Screening and eligibility
Duplicate records (n=63) were removed using WoS automatic tools. Title and abstract screening excluded 300 records that were unrelated to smoking, aging, or chronic disease. Full-text eligibility assessment removed an additional 354 records, and the final dataset included 1454 studies, which formed the basis of all bibliometric analyses. All selected studies were published in trusted, peer-reviewed journals, with no evidence of predatory or fake journals among them. All visualizations, including keyword co-occurrence maps, country collaboration networks, and bibliographic coupling structures, were produced using VOSviewer v1.6.20.
RESULTS
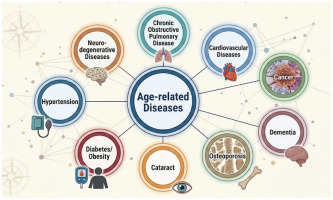
The constellation of disorders most commonly linked with late life including cardiovascular diseases, cancer, dementia, osteoporosis, cataract, diabetes/obesity, hypertension, chronic obstructive pulmonary disease and neurodegenerative diseases, are visualized in Figure 2. This figure illustrates the dominant categories of age-related conditions that form the clinical landscape into which smoking introduces additional biological stressors.
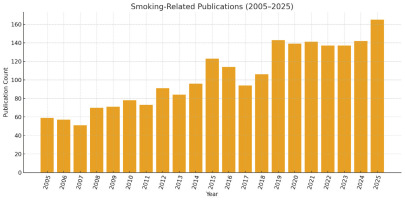
Figure 3 shows the number of publications by year, showing an upward trend in the temporal evolution of smoking-related publications between 2005 and 2025, reflecting the growing scientific interest in smoking, aging, and age-related diseases over the last two decades. Annual publication counts reveal that research activity in this domain was relatively modest during the early years of observation, with fewer than 60 publications in 2005 and 2006. This period represents a foundational stage in which smoking research was largely dominated by traditional epidemiological and clinical perspectives, and the intersection with age-related biological mechanisms had not yet become a mainstream research focus.
From 2008 onward, a gradual growth trajectory becomes visible. The rise between 2008 (70 publications) and 2012 (91 publications) corresponds to the expansion of molecular and epigenetic aging frameworks, particularly the emergence of oxidative stress and telomere biology as core explanatory models linking smoking to accelerated aging. The noticeable increase in 2015 (123 publications) represents a significant turning point. This year aligns with the widespread scientific adoption of aging biomarkers, especially the early development of epigenetic clocks, which opened new conceptual pathways for evaluating smoking-induced biological aging. Following this inflection, the 2016–2018 interval shows a temporary fluctuation, with values ranging from 94 to 114 publications. These fluctuations likely reflect evolving methodological approaches and the diversification of research subfields such as neurocognitive aging, smoking cessation in older adults, and cardiopulmonary vulnerability. Despite the variability, the underlying trajectory remains strongly positive.
The most substantial growth occurs during the 2019–2025 period. Publication counts exceed 135 each year and reach their peak in 2025 with 165 publications. This accelerating trend is consistent with global scientific interest in aging research, the integration of omics technologies, and the emerging focus on multimorbidity, frailty, and complex geriatric syndromes. The rise also coincides with increased global emphasis on healthy aging initiatives by WHO and the recognition of smoking as a modifiable accelerator of age-related disease mechanisms.
The polynomial trendline supports these observations by illustrating a non-linear but robust upward trajectory, indicating that the expansion of research is not merely incremental but structurally accelerating. The curvature of the polynomial fit suggests that smoking-related aging research is likely to continue growing, driven by advances in biological aging science, and the intensifying global burden of age-associated chronic diseases.
Figure 4 is a keyword co-occurrence map that illustrates the intellectual structure and thematic density of smoking-related research, revealing how the field has evolved around several interconnected conceptual clusters. At the center of the visualization, the dominant node ‘smoking’ anchors the entire network, reflecting its overwhelming presence in the literature and its role as the primary driver of surrounding research themes. The size of this node and the density of its linkages indicate that smoking functions not merely as a behavioral factor, but as a central axis connecting epidemiological patterns, biological mechanisms, and disease outcomes.
Immediately surrounding the core node is a prominent cluster focused on ‘tobacco’, ‘risk factors’, and ‘epidemiology’. These terms form a high-impact research stream concerned with population-level patterns, incidence, prevalence, and the identification of modifiable contributors to disease. Their strong interconnections suggest that smoking research is heavily grounded in surveillance-based methodologies and risk assessment paradigms. This aligns with broader scientific efforts to quantify the health consequences of smoking across diverse populations. Another major thematic area is represented by the cluster containing ‘lung cancer’, ‘COPD’, ‘asthma’, ‘oxidative stress’, and ‘inflammation’. This grouping reflects the well-established biological pathways linking smoking to respiratory disease and systemic physiological damage. The close proximity of ‘aging’ to these mechanistic terms suggests a growing recognition that smoking contributes to accelerated biological deterioration.
A separate but related network branch includes ‘nicotine’, ‘e-cigarettes’, and ‘chronic diseases’, indicating an expanding interest in alternative tobacco products and their long-term health implications. The connections between nicotine-related terms and aging-related mechanisms point to an emerging research direction that extends beyond traditional combustible tobacco use. Toward the right side of the map, a distinct blue cluster comprising ‘children’, ‘adolescent’, ‘health promotion’, and ‘tobacco use’ highlights a parallel research stream addressing early-life exposure and preventive interventions. Although this cluster is less directly connected to aging, its presence underscores the life-course perspective adopted in smoking research and its implications for cumulative risk in older adulthood.
Overall, the visualization demonstrates that smoking-related scholarship is organized around interdependent themes integrating epidemiology, disease mechanisms, public health, and biological aging. The map confirms that the scientific landscape naturally converges toward understanding smoking as a multifaceted determinant of long-term health trajectories, providing strong contextual grounding for aging acceleration due to smoking.
Supplementary file Figure 1 illustrates the international collaboration network in smoking-related research and reveals a highly interconnected global structure dominated by a small number of high-output countries. The United States appears as the most influential and productive node, indicated by its large cluster size and extensive linkage density. Its position at the center of the network suggests that the US not only produces the highest volume of publications but also maintains strong collaborative ties with nearly all major scientific regions, including Europe, East Asia and the Pacific. England, Canada and Australia also form prominent hubs, reflecting their sustained engagement in tobacco-related epidemiology, public health, and aging research. The strong bilateral links among these countries indicate an integrated research ecosystem that facilitates knowledge exchange and methodological convergence. European countries such as Italy, Switzerland, France, the Netherlands and Germany occupy intermediate but strategically important positions in the network. Their collaboration patterns demonstrate cross-continental interactions with both North American and Asian partners. The presence of the People’s Republic of China, India, and Japan as substantial nodes highlights the increasing involvement of Asian countries, which is consistent with their growing scientific capacity and the rising regional burden of smoking-related diseases. Türkiye, Poland, Iran and Spain appear as smaller but active contributors, connected through multi-directional collaborations rather than isolated national research lines. The chronological color gradient displayed in Supplementary file Figure 1 indicates that most collaborative intensification has occurred after 2016, suggesting a recent global shift potentially toward multi-country investigations or large-scale datasets.
DISCUSSION
This bibliometric analysis provides a comprehensive overview of the scientific landscape connecting smoking, biological aging, and age-related diseases, revealing several important aspects into how these domains interact. One of the most significant findings of this study is the consistent and strong linkage between smoking-related research and major chronic diseases of aging, including cardiovascular diseases, chronic respiratory conditions, neurodegenerative disorders, metabolic diseases, and cancers, as demonstrated in the keyword, co-citation, and collaboration networks.
These findings reinforce a critical epidemiological principle: age is a non-modifiable determinant of chronic disease, whereas smoking is one of the most powerful modifiable risk factors influencing the trajectory of aging-related morbidity. The interplay between these two forces, one inevitable, the other preventable, emerged throughout the mapped scientific literature as a central theme shaping disease progression in later life.
The bibliometric patterns observed in this study also highlight an expanding recognition of smoking as a contributor to accelerated biological aging. This aligns with recent mechanistic studies showing that cigarette smoke amplifies oxidative stress, epigenetic drift, endothelial dysfunction, telomere attrition, DNA methylation aging, and immune senescence19-21. These pathways collectively mirror, and in many cases intensify, biological processes typically attributed to normal aging. Consequently, our findings support the conceptual framing of smoking-induced acceleration of biological and pathological aging processes.
Previous systematic reviews and cohort studies have consistently shown that older smokers have significantly elevated risks of multimorbidity, frailty, cognitive decline, coronary artery disease, COPD progression, osteoporosis, and disability compared with age-matched non-smokers22,23. However, these studies typically investigate disease categories in isolation and do not explicitly incorporate biological aging science. Our findings indicate that research addressing smoking and late-life health outcomes remains fragmented across disease silos, lacking a cohesive theoretical model.
An additional important observation is the lack of focused research specifically on older adult smokers, despite their disproportionately high burden of disease. The bibliometric maps reveal substantial activity in general smoking epidemiology and youth/early adulthood prevention, yet relatively fewer publications concentrate on geriatric populations. This represents a critical gap because the physiological vulnerabilities of older adults reduced regenerative capacity, immune senescence, accumulated oxidative burden render them especially susceptible to smoking’s harmful effects24. Several studies demonstrate that elderly individuals who smoke experience steeper functional decline and are at markedly increased risk of hospitalization, disability, and premature mortality compared with non-smoking peers25.
The international collaboration patterns identified in this study offer further information with policy implications. Countries such as the US, England, Canada, Australia, China, and several European nations appear as central nodes in global research coordination. These nations lead in producing high-impact publications and forming multi-country networks capable of advancing mechanistic, clinical, and epidemiological knowledge.
Strengths and limitations
The strengths of this study include its large dataset drawn from a high-quality bibliographical source (Web of Science), rigorous screening, and exclusive reliance on VOSviewer for standardized bibliometric visualization. The integration of co-occurrence, co-citation, and collaboration maps strengthens the reliability of thematic interpretations and provides a multifaceted perspective on the field’s evolution.
However, several limitations should be acknowledged. First, the study is limited to WoS-indexed publications; important literature in Scopus, PubMed, or regional databases may not be captured. Second, bibliometric analyses depend on metadata rather than full-text content, which may omit nuance in mechanistic or clinical findings. Third, citation-based metrics favor older publications and output from high-income countries, potentially underrepresenting emerging research from low- and middle-income regions. Fourth, because this study relies on bibliometric methods rather than empirical clinical data, causal relationships between smoking, aging, and disease pathways cannot be inferred.
CONCLUSIONS
This study provides a comprehensive bibliometric mapping of smoking and aging research as a framework for understanding how tobacco exposure accelerates age-related disease processes, revealing a highly interconnected research field characterized by shared mechanisms, overlapping disease pathways and growing global interest.